- Industry Perspective – March 2026
- Outokumpu Advert
- Sassda News : Strategic Session Charts Export – Led Path for Stainless Steel Growth Sector
- Fastenright Advert
- GPS Round Up
- State of the Stainless Steel Nation
- Technical Case Study – The Visual Science of Stainless Steel in Architecture
- Professional Profile : Bjon Frljak
- Case Study: Stainless Steel Corrodes More Slowly than Carbon Steel But by How Much?
- Case Study: How Temperature and Thermal Expansion Affect Stainless Steel Cladding Design
- Sassda News: From Local Limits to Global Markets
- Africa Market Intelligence – Kenya
- Member News
- Obituary – Lesley Squires
- Sassda News: Eastern Cape Golf Day

Corrosion is a global scourge that destroys manmade materials. This includes concrete, metals, plastics, and even composite materials. Studies indicate that half the world’s steel production is used to repair corroded steel structures.
Sassda regularly receives questions about the corrosion rate of stainless steel compared to carbon steel. This is a valid question and suggests growing interest in life cycle costing, sustainability, and environmental impact.
However, the question may appear straightforward, but the answer is complex.
Unlike carbon steel, stainless steel is a family of materials, not a single alloy or even a small group of closely related alloys. It can be grouped into ferritic, martensitic, austenitic, duplex, and precipitation-hardening stainless steels, each representing a branch on the family tree. Within these five groupings are more than 200 different alloys, each developed for a specific purpose and application.
The chemical composition of these grades, together with their intended application, determines their corrosion rate. That rate varies significantly across the alloy spectrum. It is therefore difficult to make direct comparisons, as corrosion rates vary widely depending on the environment.
Industry typically relies on representative performance data supported by published corrosion resistance comparisons rather than single numerical values, which depend on factors such as chemical composition, pH, chloride concentration, temperature, and oxygen availability.
Why Stainless Steel Corrodes More Slowly Than Carbon Steel?
Stainless steels contain at least 10.5% chromium. This chromium forms a thin, self-healing passive oxide film that dramatically slows corrosion. Carbon steel lacks this passive film and rusts readily. It is generally classified as having low corrosion resistance and will rust in the presence of moisture, acids, or chlorides unless protected by coatings.
Relative Corrosion Performance (Qualitative Ranking) in mildly corrosive or atmospheric conditions Information from BSSA and Outokumpu implies that the following qualitative ranking for stainless steel exists when comparing it to carbon steel. The table shows a practical ranking from fastest corrosion to slowest corrosion under typical atmospheric or mildly aggressive aqueous conditions.
Typical Relative Corrosion Rate Ranges (General Guidance)
If carbon steel is compared to different stainless steel grades in different environments the following qualitative ranges are commonly accepted in corrosion engineering and reflected in stainless steel selection tables.
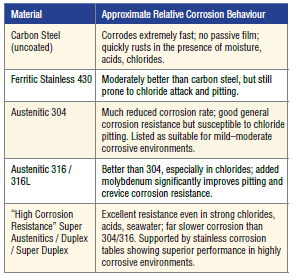
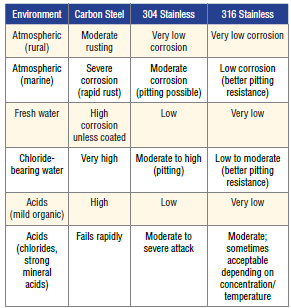
Corrosion tables (Alleima, Outokumpu) show that carbon steel corrodes quickly in nearly all aqueous acidic and chloride environments. Stainless steel grade 304 is acceptable in many mildly corrosive environments but becomes susceptible in chlorides. The molybdenum containing grade 316 shows significantly better performance due to the molybdenum and is recommended for seawater, chlorides, and industrial chemicals.
Highly alloyed stainless steels such as the super austenitics, and duplex/super duplex grades function excel in severe chemicals and high chloride solutions.
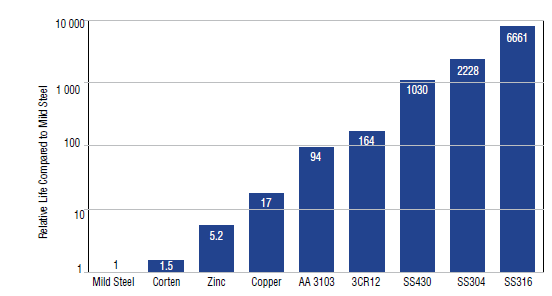
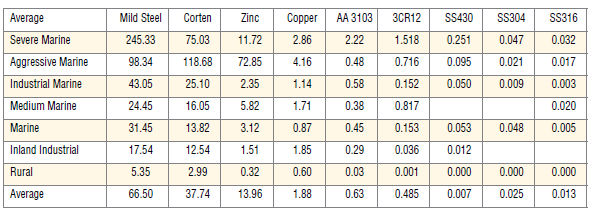
Theoretical comparisons
This graph shows the relative life span of various materials compared under the same functional conditions. Carbon steel is the base for the comparison. As an example, this would indicate that in conditions where mild steel would last one year, grade 3CR12 would last an estimated 164 years.
The table shows the result of corrosion tests done on various materials under various corrosive conditions. The bottom row shows the average over the range of conditions. For example, under severe marine conditions, grade 316 will suffer a material loss of 0.032% per year whilst 3CR12 will an
estimated 1.518% loss over the same period.
Conclusion
It is important to ensure that that when working with stainless steel grade selection, the application and the operational conditions are well defined. This will assist in selecting the grades that might be suitable for the application. Corrosion rates on stainless steel are determined by alloy composition, surface finish and condition, availability of environmental oxygen, level of chlorides, solution strength and temperature. When all the information on the application is available Iso-corrosion diagrams can be used to narrow down the list of potential suitable grades. It is then advisable to do a physical test on samples of the shortlist of alloys under the operational conditions to make a final decision.

